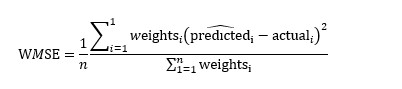At what temperature is the \"effective\" speed of gaseous hydrogen molecules (molecular weight = 2) equal to that of oxygen molecules (molecular weight = 32) at 47^∘C ?

PDF) Examination of the Weighted Root Mean Square Residual: Evidence for Trustworthiness | Dexin Shi - Academia.edu
![PDF] Weighted root mean square approach to select the optimal smoothness parameter of the variational optical flow algorithms | Semantic Scholar PDF] Weighted root mean square approach to select the optimal smoothness parameter of the variational optical flow algorithms | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/a62de7de4f42d5122a39e769ccfdbae95297972e/5-Figure1-1.png)
PDF] Weighted root mean square approach to select the optimal smoothness parameter of the variational optical flow algorithms | Semantic Scholar

The relationship between weighted root-mean-square acceleration value... | Download Scientific Diagram
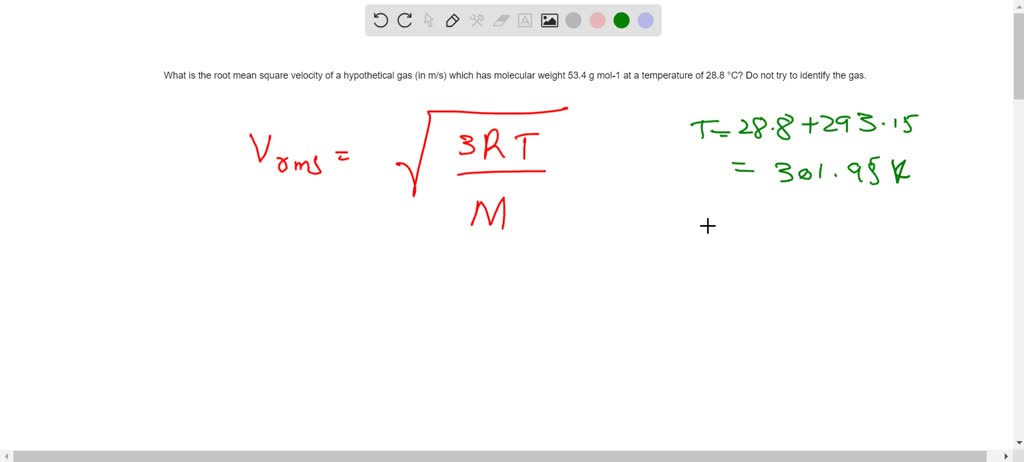
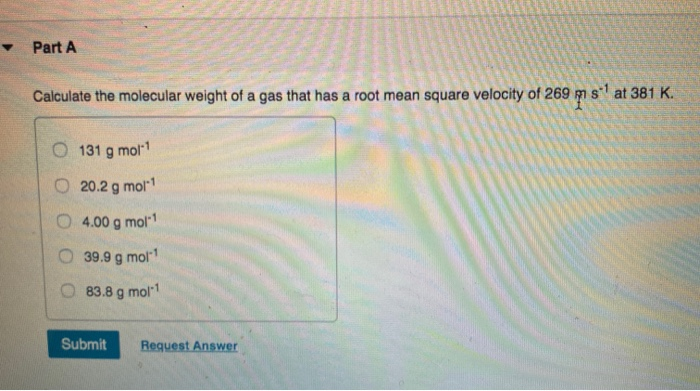
SOLVED: What is the root mean square velocity of a hypothetical gas (in m/s) which has molecular weight 53.4 g mol-1 at a temperature of 28.8 °C? Do not try to identify

The molecular weight of a is 2. Calculate the root mean square velocity of its molecules at 0^(@)C and 100^(@)C givenn that R=8.3Jmol^(-1)K^(-1)

At `400 K`, the root mean square (rms) speed of a gas X (molecular weight = 40) is equal to the most - YouTube
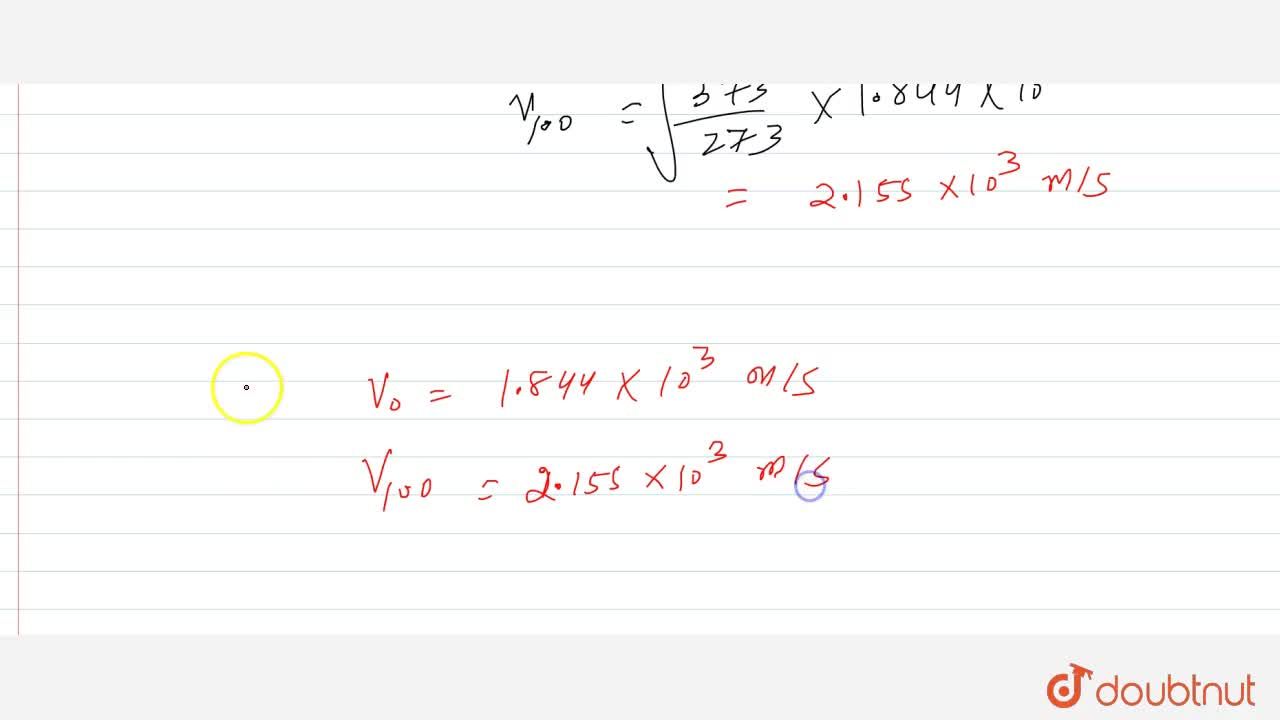


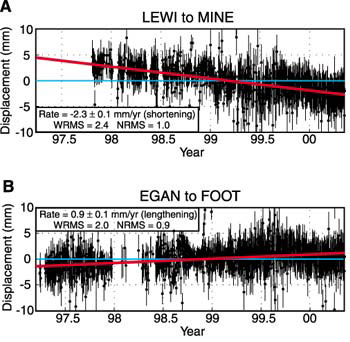



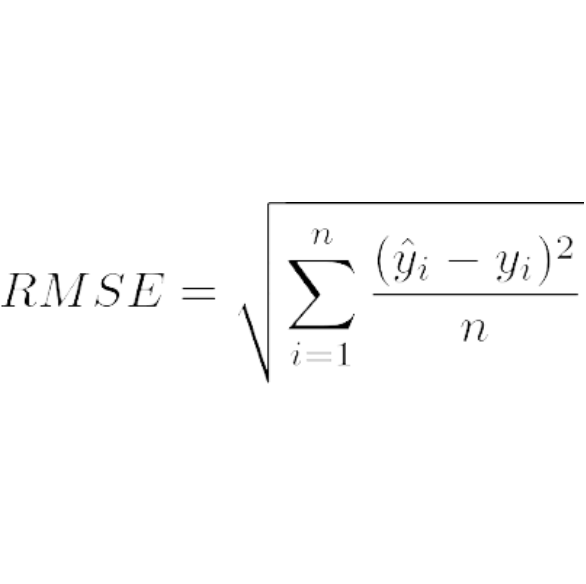



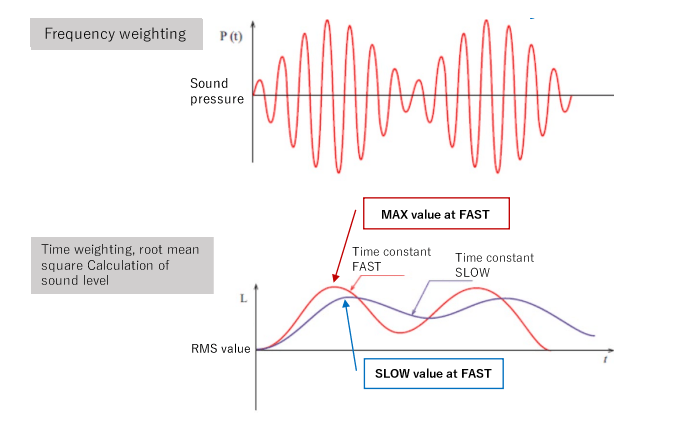
![Prediction errors [weighted root mean squared error (wRMSE)] of... | Download Scientific Diagram Prediction errors [weighted root mean squared error (wRMSE)] of... | Download Scientific Diagram](https://www.researchgate.net/publication/323859153/figure/fig4/AS:606066937171968@1521509026444/Prediction-errors-weighted-root-mean-squared-error-wRMSE-of-individual-regression.png)